ATP: What is triphosphate (ATP)?
Last Update on 15 November 2022
| Introduction | How does body make ATP? | Functions of ATP| Mechanism of ATP |Test to measure ATP | Medical Signification of ATP |
1. Introduction:
The body requires energy since it is a complex organism and needs it to continue functioning. Every physiological action that needs energy is impacted by adenosine triphosphate ATP, which is present in every cell. Once released into the extracellular environment, ATP can transmit messages between cells.
Adenosine Triphosphate ATP improves body composition, increases strength and power, and lessens fatigue. Supplementing with ATP enhances cardiovascular health. The movement of macro-molecules like proteins and lipids into and out of the cell depends heavily on ATP. The energy needed for active transport mechanisms to move such molecules over a concentration gradient is provided by the hydrolysis of ATP.
Adenosine triphosphate is utilized and saved as energy at the cellular level (ATP). The nucleoside triphosphate ATP is composed of adenine, ribose, and 3 sequentially bonded phosphate groups.
ATP is often referred to as the “energy currency” of the cell since it saves easily accessible energy in the link between the second and third phosphate groups. In addition to providing energy, the hydrolysis of ATP supports a variety of cellular processes, such as DNA/RNA synthesis and signaling. Numerous catabolic processes, including cellular respiration, beta-oxidation, and ketosis, provide energy for the synthesis of ATP.
Most ATP synthesis takes place during cell respiration inside the mitochondrial matrix, producing about 32 ATP molecules for every glucose molecule that is oxidized. A few of the activities that employ ATP as an energy source include ion transport, muscle function, nerve impulse transmission, enzyme activation, and chemical synthesis. ATP is crucial for both of these activities as well as other ones. As a response, 100 to 150 moles of ATP must be hydrolyzed daily in order for cells in the human body to function properly. In the sections that follow, we’ll look more closely at ATP’s function as a crucial chemical in the normal functioning of the cell.
2. How does the human body make ATP?
In the sections that follow, we’ll look more closely at ATP’s function as a crucial chemical in the normal functioning of the cell. Most of the adenosine triphosphate in cells is produced by the enzyme ATP synthase, which does this by transforming ADP and phosphate to ATP. The ATP molecule breaks down one of its three phosphates when a cell has to expend energy to complete a task, resulting in the formation of ADP (Adenosine di-phosphate) + phosphate. The energy that was tied to that phosphate molecule is now free to be utilized by the cell to carry out its functions.
3. The functions of Triphosphate ATP:
Numerous essential biological and cellular processes, including ATP hydrolysis, demand energy. Active transport, Purinergic signaling, synaptic signaling, intracellular signaling, DNA and RNA synthesis, and muscle contraction are a few of them. Although not a complete list, these subjects cover some of the vital areas that adenosine triphosphate plays.
3.1. ATP in Intracellular Signaling:
On ATP, signal transduction largely depends. Kinases, the most prevalent ATP-binding protein, can utilize ATP as a substrate. A kinase’s phosphorylation of a protein can initiate a signaling cascade, which can then influence a number of intracellular signaling pathways. Kinase activity must be tightly managed since it is crucial to the functioning of the cell. The presence of the magnesium ion helps to regulate kinase activity. Magnesium ions, which are bound at the phosphate oxygen centers and are present in the cell along with ATP, play a role in regulation.
In addition to kinase activity, it can work as a typical trigger of intracellular messenger release. Among these messengers include hormones, various enzymes, lipid mediators, neurotransmitters, nitric oxide, growth regulators, and reactive oxygen species. Adenylate cyclase is an example of an intracellular signaling enzyme that employs ATP as a substrate. The majority of the time, this mechanism involves G-protein-linked receptor signaling pathways.
The process by which ATP is transformed by adenylate cyclase into cyclic AMP aids in the release of calcium from intracellular stores. Activating protein kinases, acting as a secondary messenger in hormone signaling cascades, and regulating ion channel performance are some of the additional tasks carried out by cAMP.
3.2. DNA/RNA Synthesis:
The production of DNA and RNA needs ATP. One of the four nucleotide-triphosphate monomers demanded RNA synthesis is ATP. Similar mechanisms are used in DNA synthesis, except in DNA synthesis, the ATP is first changed by withdrawing an oxygen atom from the sugar to produce deoxyribonucleotide or dATP.
3.3. Purinergic Signaling:
A type of extracellular paracrine signaling known as purinergic signaling is carried out by purine nucleotides, such as ATP. This procedure frequently involves the activation of purinergic receptors on nearby cells, which results in the transmission of signals to control intracellular processes. IP3 and other typical exocytotic regulatory systems control the release of ATP from vesicular storage. It is further evidence that ATP is a crucial mediator of purinergic neurotransmission in both sympathetic and parasympathetic neurons because other neurotransmitters are co-stored and co-released with ATP. The purinergic responses that ATP can trigger include those that regulate pain, neuronal-glial connections, autonomic processes, and vascular tone.
3.4. Neurotransmission:
The largest consumer of ATP in the body, the brain consumes around 25% of the body’s total energy. An important source of energy for proper synaptic transmission and neuronal signaling is controlling ion concentrations. Synaptic transmission requires a lot of energy. In order to produce ion gradients that carry neurotransmitters into vesicles and prepare the vesicles for exocytosis, ATP is required at the presynaptic terminal.
Neuronal signaling requires action potentials that indicate the discharge of loaded vesicles at the presynaptic terminal. A new signal cannot be sent until ATP has restored the ion concentration in the axon following each action potential active transport is in charge of restoring the sodium and potassium ion concentrations to their starting values following an action potential through the Na/K ATPase. Throughout this process, which also requires the passage of three sodium ions and two potassium ions against the gradient of concentration as they leave and enter the cell, accordingly, one ATP molecule is hydrolyzed.
Action potentials that move through the axon and reach the presynaptic terminal initiate vesicular release. Ion gradients are produced by the action potentials, which then travel along the axon across depolarization of the axon and transmit a signal in the location of the terminal. Only around one billion sodium ions can disseminate a single action perspective. To restore the sodium/potassium ion concentration after each discharge, neurons will be required to hydrolyze roughly one billion ATP molecules.
The majority of the gray matter volume of the brain is made up of excitatory connections. To stimulate postsynaptic excitatory glutaminergic receptors, glutamate-containing vesicles will be released into the synaptic cleft. Because there are around 4,000 glutamate molecules packed into one vesicle, loading these molecules demands a lot of ATP. Large energy reserves are required to start the vesicle’s release, power the glutamatergic postsynaptic processes, and recycle both the vesicle and any leftover glutamate. Therefore, mitochondria are close to glutamatergic vesicles due to the substantial amounts of energy needed for glutamate packing.
3.5. ATP in Muscle Contraction:
Muscle contraction would not be possible without ATP, making it a vital biological process. The three basic ways that adenosine triphosphate contributes to muscular contraction are as follows. In the first technique, myosin cross-bridges cycle while exerting force against neighboring actin filaments. In the second, calcium ions from the myoplasm are actively transported throughout the sarcoplasmic reticulum in defiance of concentration gradients. The active movement of sodium and potassium ions across the sarcolemma allows calcium ions to be released when an input is received. This is ATP’s third assignment. The hydrolysis of adenosine triphosphate fuels each of these processes.
4. Mechanism of Triphosphate (ATP):
The body can manufacture adenosine triphosphate in a number of ways, depending on the current metabolic state. When oxygen is available, cell respiration, beta-oxidation, ketosis, lipid, and protein breakdown can all lead to the creation of ATP in response to anaerobic circumstances.
4.1. Cellular Respiration:
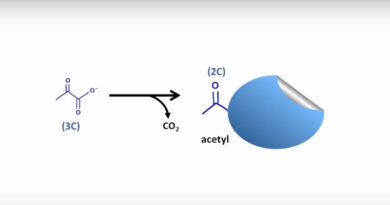
Catabolizing glucose into acetyl-CoA during cellular respiration produces a high-energy electron transport chain that will be oxidized during oxidative phosphorylation to produce ATP. During glycolysis, the initial phase of cell respiration, one glucose molecule divides into two pyruvate molecules. In this step, the substrate is phosphorylated by the enzymes PFK1 and pyruvate kinase, resulting in the production of two adenosine triphosphate. Two reduced NADH electron transport system molecules are also produced.
The pyruvate dehydrogenase complex then oxidizes the pyruvate molecules to produce an acetyl-CoA molecule. The citric acid cycle then completely oxidizes the acetyl-CoA molecule to produce carbon dioxide and reduced electron carriers. Two molecules of carbon dioxide, one equal of ATP, 3 molecules of NADH, and one molecule of FADH2 are the end products of the citric acid cycle.
The hydrogen ions (protons) from the mitochondrial matrix are subsequently moved against their gradients into the inner membrane space by these high-energy electron carriers, where they are then transmitted to the electron transport chain. Then, ATP synthase generates ATP molecules as protons flow down the electrochemical gradient. Various amounts of ATP are produced, according to the electron carrier that supplied the protons. One NADH molecule generates two and a half ATP molecules, compared to one and a half produced by FADH2.
4.2. Beta-Oxidation:
By using beta-oxidation, organisms can produce ATP in another way. During beta-oxidation, fatty acid chains irreversibly shorten to form Acetyl-CoA molecules. Throughout each cycle of beta-oxidation, the fatty acid is decreased by two carbon lengths, producing one molecule of acetyl-CoA, which can be oxidized in the citric acid cycle, as well as one molecule of each NADH and FADH2, which contributes a high-energy electron to the transport process.
4.3. Ketosis:
The ketosis mechanism generates ATP by breaking down ketone compounds. During the catabolic activity of ketone bodies during ketosis, 22 ATP molecules and 2 GTP molecules are created for each acetoacetate molecule that is oxidized in the mitochondria.
4.4. Anaerobic Respiration:
If there is not enough or no oxygen available for cellular respiration, cells may participate in anaerobic respiration. An accumulation of NADH molecules results from the inability of anaerobic cells to metabolize NADH to NAD+, which limits GAPDH activity and glucose uptake. Pyruvate is transformed to lactate in the lactic fermentation process, which occurs in the oxidation of one NADH molecule, in order to keep homeostatic levels of NADH. The two NADH molecules produced during glycolysis are oxidized during lactic fermentation in order to keep the NAD+ reservoir full. In this mechanism, each glucose molecule yields just two adenosine triphosphate molecules.
5. Tests to Measure Triphosphate (ATP) Level:
There are many different ways to determine intracellular ATP levels. It is a widely accepted approach to use firefly luciferase, an enzyme that causes luciferin to be oxidized. The energy output of this process, which releases a measurable photon of light known as bioluminescence, makes it quantifiable.
6. Medical Signification of Triphosphate (ATP):
6.1. ATPs Role in Pain Control:
Acute perioperative discomfort has decreased, according to clinical trials on ATP. In these investigations, patients received intravenous adenosine triphosphate. Adenosine is administered intravenously, where it interacts with the A1 adenosine receptor and starts a signaling cascade that eventually helps inflammation-related pain relief. Studies have shown that adenosine compounds, when administered in moderate quantities, can reduce allodynia and hyperalgesia. A1 adenosine receptor activation produces effective pain relief because it has a slow onset and a long half-life, sometimes lasting for weeks.
6.2. Anesthesia:
During anesthesia, ATP supplements provided beneficial results. Evidence suggests that low doses of adenosine can relieve pain in ways similar to morphine, including neuropathic pain, ischemic pain, and hyperalgesia. Additionally, adenosine decreased the need for postoperative opioids, suggesting that the A1 adenosine receptor may have been activated for an extended period of time.
6.3. Cardiology and Surgery:
ATP is a proven and secure pulmonary vasodilator in patients with pulmonary hypertension. During surgery, a patient’s blood pressure can also be lowered with the use of adenosine and ATP.